A Next-Generation Oncolytic Immunotherapy Engineered for Precision, Power, and Safety
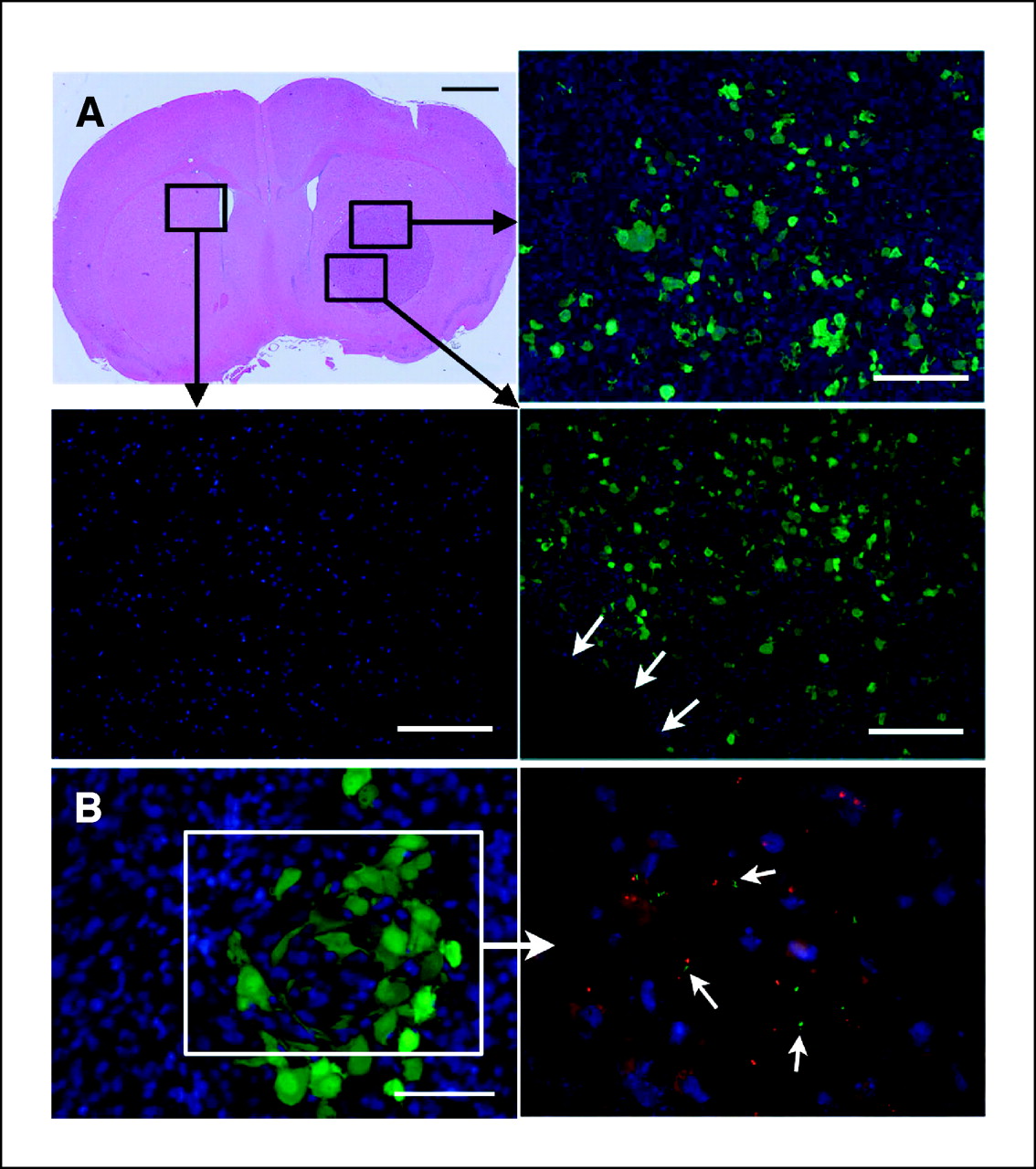
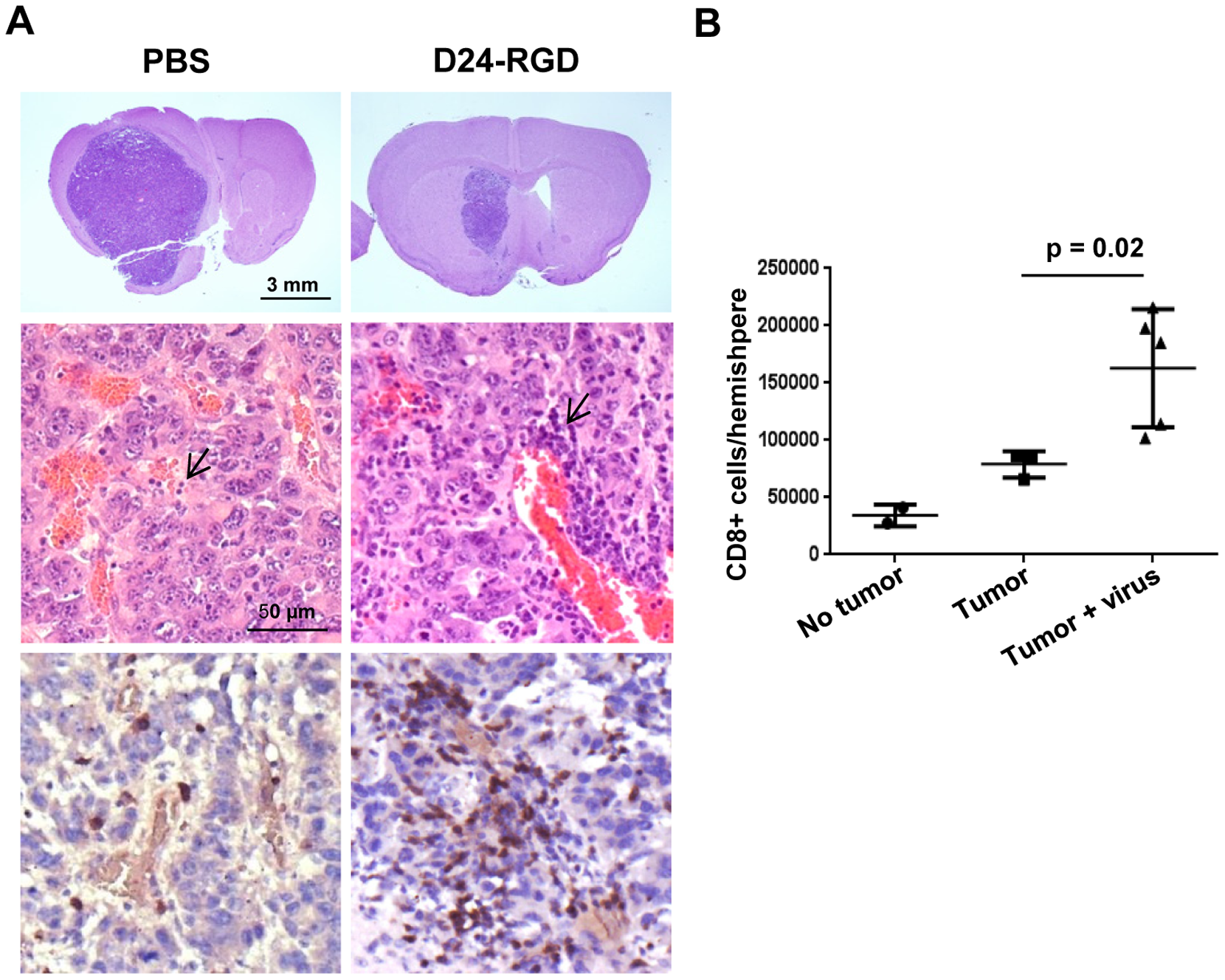
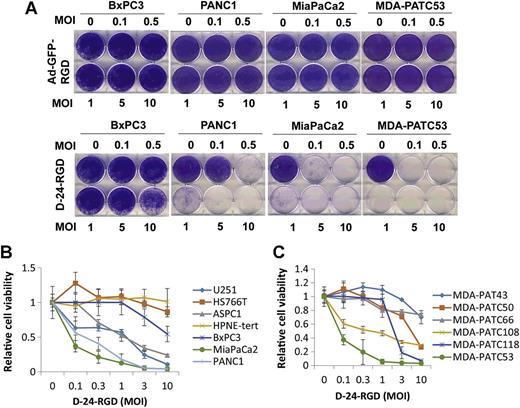
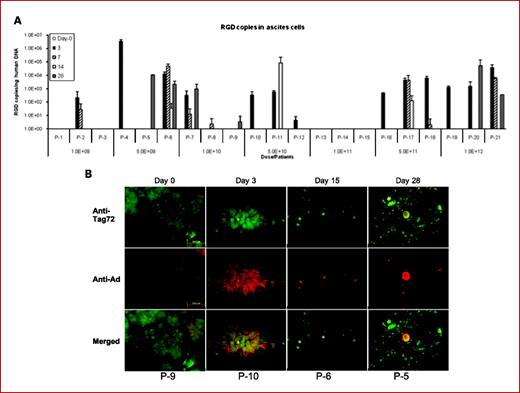
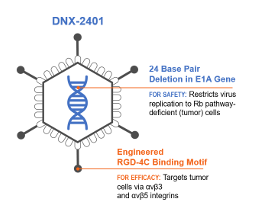
DNX-2401 is an oncolytic immunotherapy designed to fulfill the dual requirements of high potency and safety. To accomplish this, two stable genetic changes in the adenovirus genome were engineered that cause it to replicate selectively in retinoblastoma (Rb) pathway-deficient cells and infect tumor cells efficiently. Results from preclinical and clinical studies indicate that DNX-2401 (1) replicates in human tumors, (2) elicits tumor necrosis, (3) triggers intratumoral immune cell infiltration, and (4) can lead to long-term tumor destruction.
Structure of DNX-2401
Mechanism of Action of DNX-2401

Completed Clinical Studies
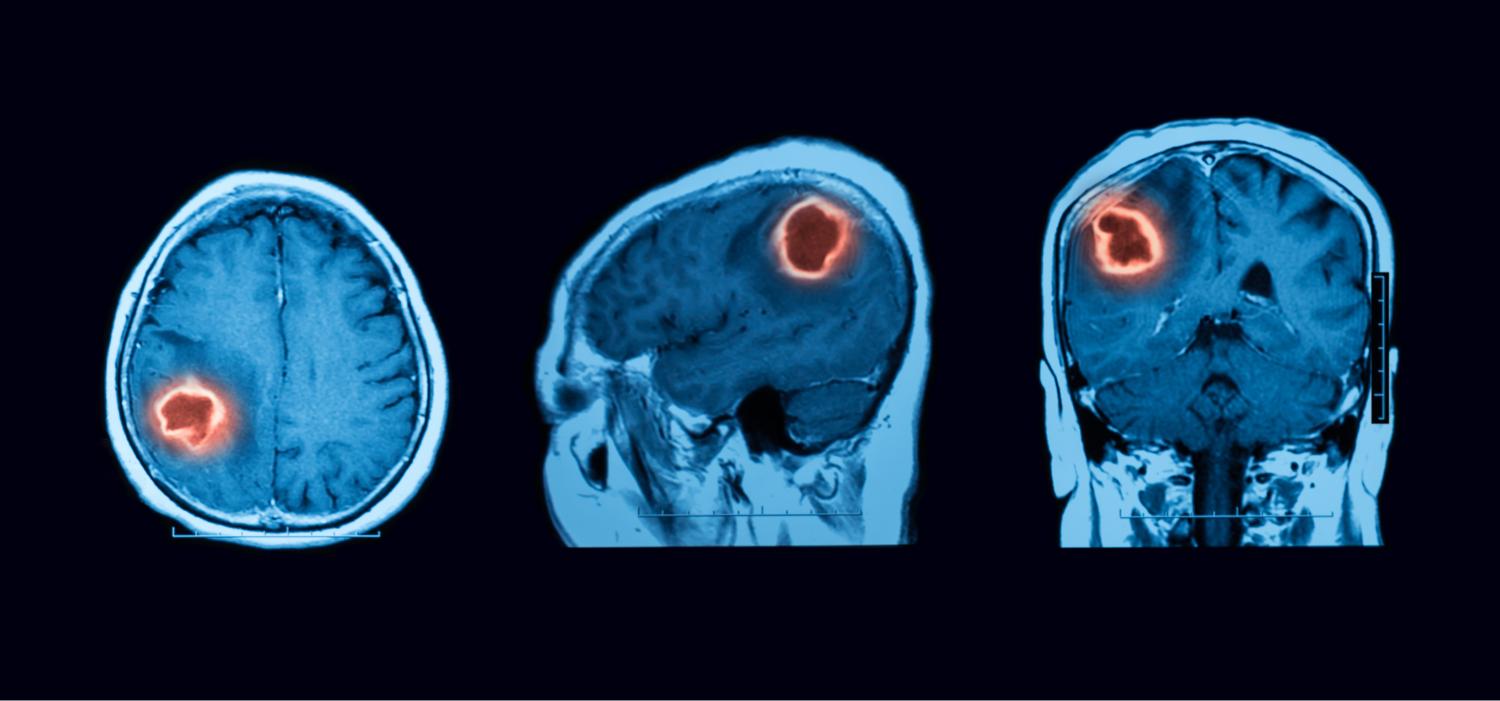
A Phase 1 trial was completed to evaluate the safety and efficacy of DNX-2401 in pediatric patients with Diffuse Intrinsic Pontine Glioma (DIPG). Patients who are less than 18 years old with newly diagnosed DIPG were eligible.
The full results of this study are published here [Gállego Pérez-Larraya 2022 ].

A Phase 1 trial was completed evaluating DNX-2401 as a monotherapy treatment for adults with recurrent glioblastoma (GBM). The full results of this study are published here [Lang 2018].

The Phase 2 CAPTIVE trial was conducted in collaboration with Merck to evaluate the efficacy and safety of DNX-2401 in combination with pembrolizumab (KEYTRUDA), Merck’s anti-PD-1 therapy. Adult subjects diagnosed with GBM or gliosarcoma who experienced disease progression after initial treatment were eligible.
The full results of this study are published here [Nassiri 2023].

$10.8 Million CPRIT Grant Award
The Cancer Prevention & Research Institute of Texas (CPRIT) provided $10.8 million grant to advance the development and commercialization of an innovative oncolytic adenovirus therapy for glioblastoma (GB), one of the most aggressive brain cancers.