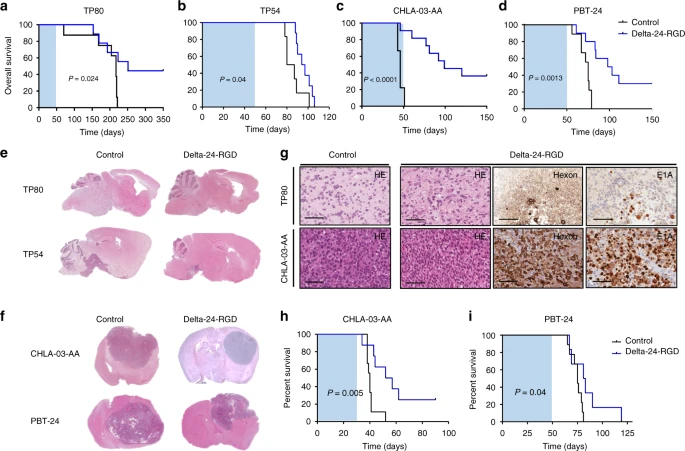
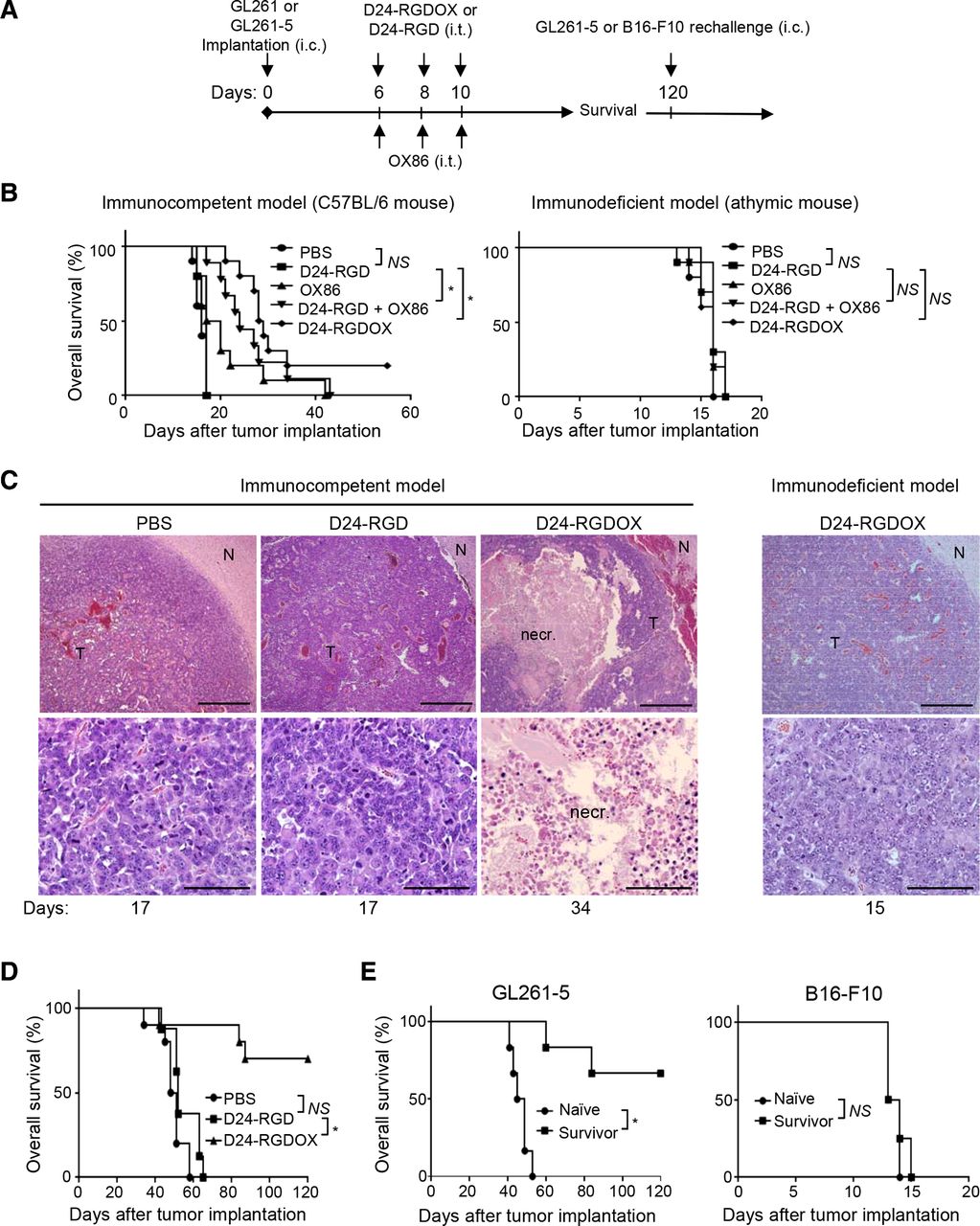
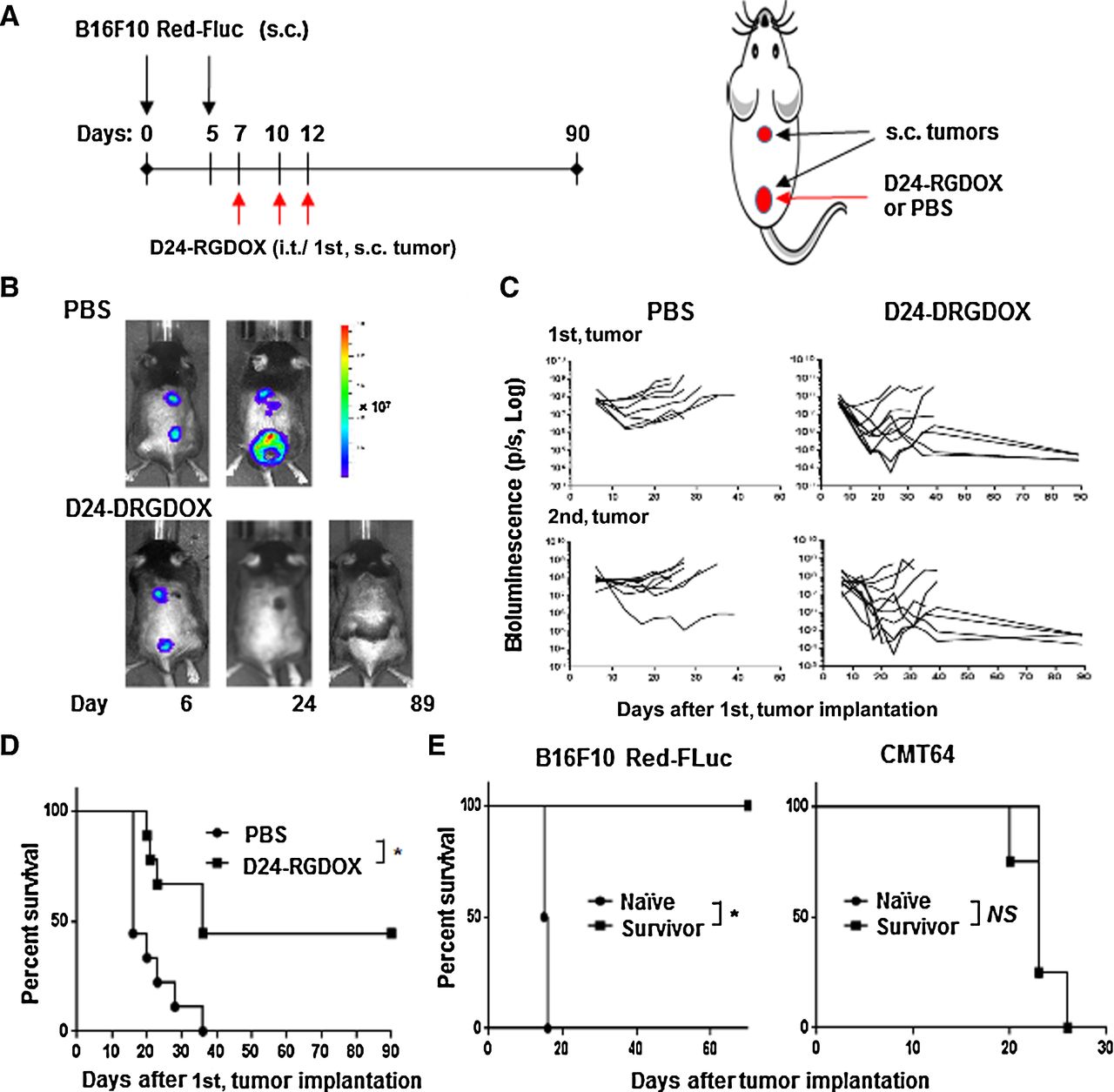
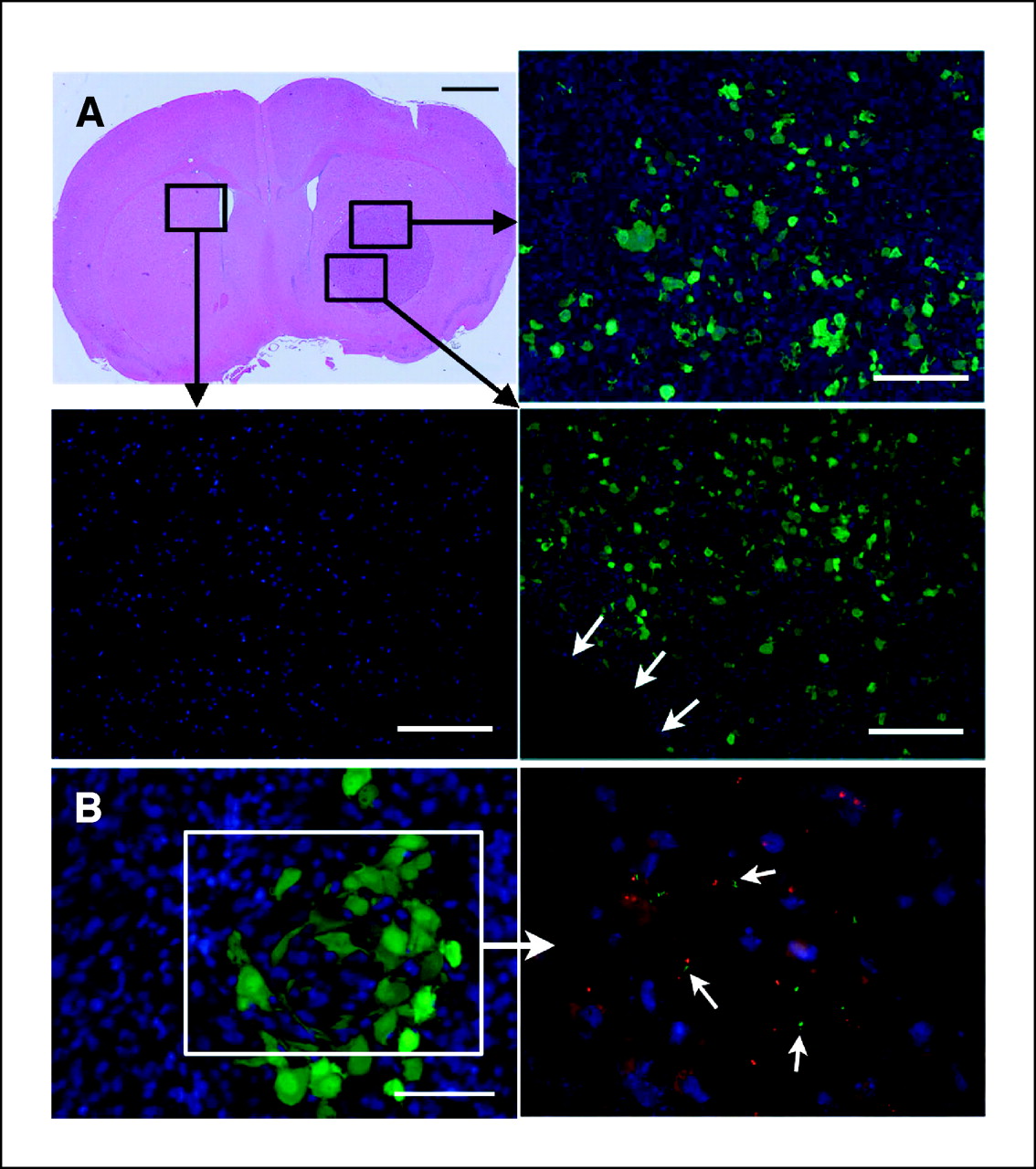
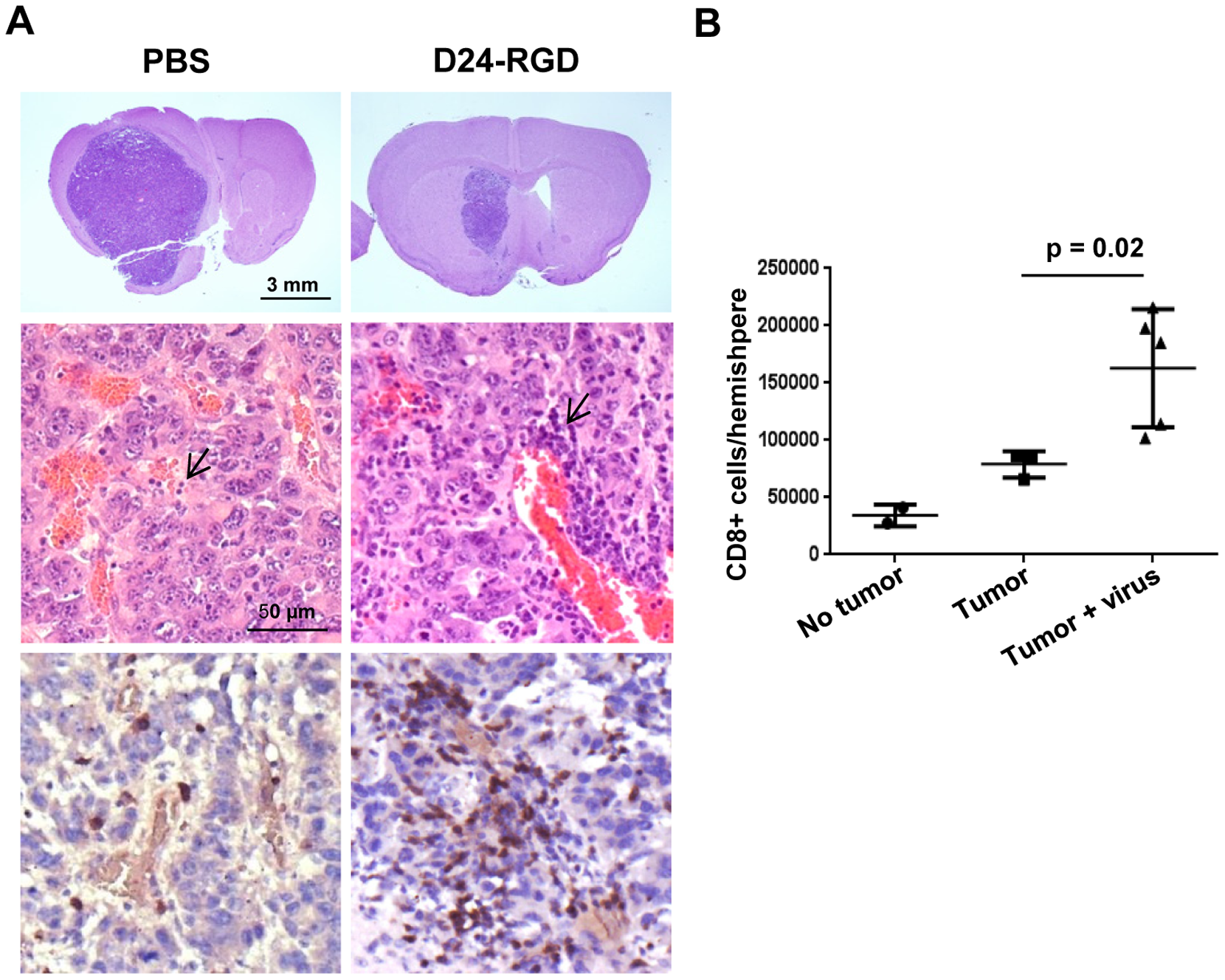
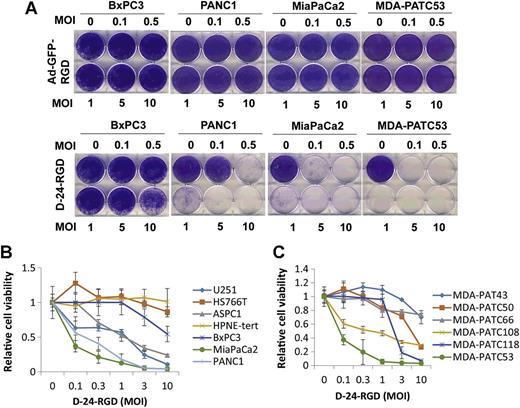
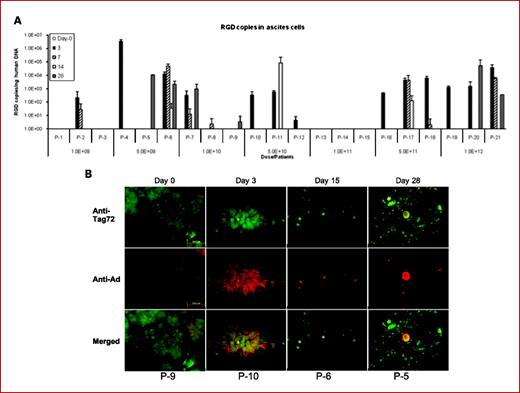
New Clinical Trials
Multi-dose DNX-2401 for Pediatric Patients with DMG
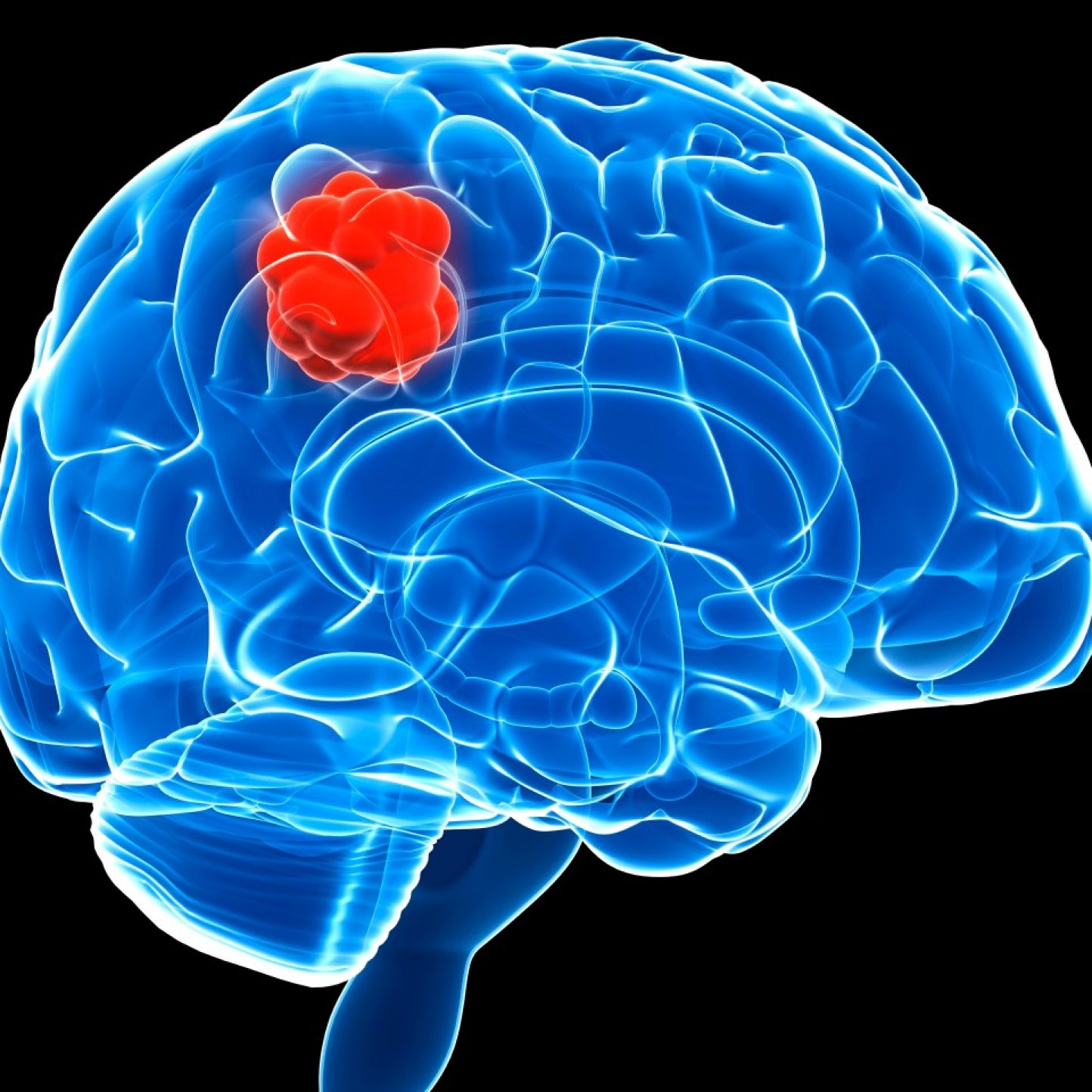
A Phase 1/2 trial is planned to evaluate the efficacy and safety of DNX-2401 in children and young adults with Diffuse Midline Glioma (DMG), including DIPG. Subjects ages 1-35 years old may be eligible.

DNX-2401 for Pediatric Patients with Recurrent and Refractory High Grade Brain Tumors
A Phase 2 trial is planned to evaluate the efficacy and safety of multiple doses of DNX-2401 in children and young adults with recurrent or refractory high-grade brain tumors. Patients with several types of rare brain tumors (high-grade gliomas, DMG, ependymomas, embryonal tumors including medulloblastomas and AT/RT), ages 1-25 years old, may be eligible.